Miscarriages can be PreventedPreventing MiscarriagesAn unexpected miscarriage can shatter dreams. Two or more can be devastating. But now there is hope, and a solution. We offer a proven, painless, highly sophisticated diagnosis and treatment method. By combining cell flow cytometry and immune enhancing vaccinations, our success rate is 80 percent. To date, we have had hundreds of births. Reproductive Immunology ExplainedOne in every 200 couples are too genetically similar to achieve successful pregnancy. And usually, they don't know it. That's why early detection is vital. Without intervention, the painful pattern of miscarriage occurs again and again. As the United States' only private physician group capable of performing all of the laboratory tests necessary to diagnose this condition, we can provide complete evaluation within 10 days. And we closely monitor each pregnancy, consulting with referring physicians every step of the way. Understanding Reproductive Immunology:
Causes of MiscarriagesOne in two hundred couples will experience two or more consecutive miscarriages. There are five reasons for miscarriage which have been identified:
Some women have multiple reasons for miscarriages. At Reproductive Immunology Associates we evaluate patients for immune related miscarriages. Your obstetrician will test for most other causes of pregnancy loss. The Immune SystemAdvances in immunology, the study of the body's defense systems, enable us to understand how during pregnancy, the mother's immune system is altered so that the fetus is not rejected by her body and allows the fetus to grow. The immune system is comprised of white blood cells, also known as leukocytes, which make a variety of antibodies. Some of the antibodies protect us and others are harmful to our bodies. Some of the immune issues that are important to the reproductive system are:
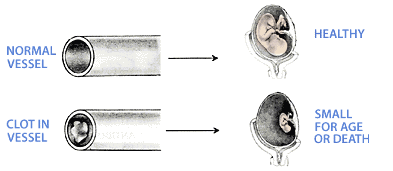
When the immune system is the cause of miscarriage, the chances of mother having a successful pregnancy without treatment after three miscarriages is 30%, after four miscarriages 25%, and after 5 miscarriages 5%. Antiphospholipid AntibodiesPhospholipid molecules are normal components of all cell membranes. Some also have glue like properties and allow cells to fuse (as you will see later). Antibodies to phospholipid molecules can, therefore, cause problems. Specifically, they can damage the inside of the blood vessel wall. This allows blood cells to stick to the site of the injury and cause blood clots.
Some phospholipid molecules have adhesion properties i.e. glue like, and allow cells to fuse. The formation of the normal placenta involves the fusion of small cells called cytotrophoblasts into giant cells known as syncytiotrophoblasts. The syncytiotrophoblasts play a key role in the regulation of nutrients going to the baby. 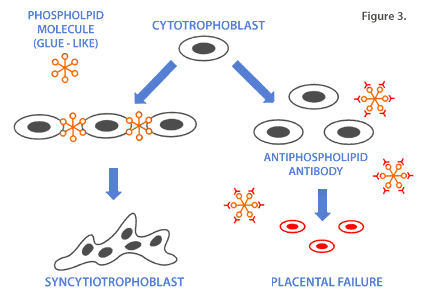
With each pregnancy loss, there is a 10% chance that the mother will develop an antibody to a phospholipid molecule. Most women with antiphospholipid antibodies are not sick. However, some have underlying autoimmune tendencies and should be appropriately evaluated. Women with underlying autoimmune diseases may have antiphospholipid antibodies even before they ever become pregnant. Treatment for Antiphospholipid AntibodiesAntiphospholipid antibodies are treated with low dose (baby) aspirin and a blood thinner called Heparin. Heparin is a very large molecule and is unable to cross the placenta. Aspirin is able to cross the placenta but the dose used is so small that the fetus is unaffected. The effectiveness of treatment is much greater when the medication, if indicated, is started prior to conception and continued throughout the pregnancy. All medication, if indicated should be discussed with one's physician. Antinuclear AntibodiesThe nucleus is the " brain " of the cell. It contains the information that regulates the function of the cell. Some people have antibodies to different nuclear components. What causes these antibodies to be made is currently under investigation but there appears to be a genetic susceptibility which may be reflected by the HLA tissue type (refer back to the section on blocking antibodies). The disease that we typically associate with antinuclear antibodies is Systemic Lupus Erythematosus (SLE). The miscarriage rate in SLE patients is much higher than that of the general population. Although most women who suffer recurrent miscarriages do not have clinical signs of SLE, many exhibit autoimmune phenomena which is similar to that seen in SLE patients. The placentas in these women are inflamed and weakened.  Patients who develop new autoantibodies like antinuclear and antiphospholipid antibodies during pregnancy have a more guarded prognosis. Treatment of Antinuclear AntibodiesWomen with ANA are treated with prednisone, a corticosteroid, which suppresses the inflammatory process and stabilizes the cell. Prednisone does not pass through the placenta easily and is also broken down by enzymes in the placenta so that the fetus is exposed to only trace amounts. Additionally, the body produces the equivalent of 8 mg per day of this corticosteroid. When indicated, Prednisone should be started prior to conception. Antithyroid AntibodiesIn 1990, Stagnaro-Green demonstrated in a prospective analysis that thyroid antibodies were markers for "at-risk" pregnancies. The two antibodies studied, anti-thyroid peroxidase and anti-thyroglobulin antibodies, are collectively referred to as anti-thyroid antibodies (ATA). Many reports have since corroborated the markedly increased prevalence of ATA in women who experience reproductive failure, especially first trimester miscarriages. Pratt, et. al., showed that 67% of women with recurrent first trimester losses had ATA, compared to 17% of controls. None of the participants in either group had clinical manifestations of thyroid disease. Although there is a highly positive correlation between the presence of ATA and fetal loss, no definitive pathophysiology has been identified. Several hypotheses have been proposed to explain this phenomenon. One hypothesis states that these patients have very mild hypothyroidism. Studies to date fail to indicate low thyroid hormone levels in those who miscarried. Proponents suggest that serum hormone levels do not necessarily reflect thyroid dysfunction. Another opinion is that ATA are markers for predisposition to autoimmune disease, and that the latter is what actually causes the miscarriage. Notable is that ATA is present in up to 45% of patients with systemic lupus erythematosus (SLE). In another study, 70.8% of patients with recurrent spontaneous abortion (RSA) had various autoantibodies, leading the authors to conclude that some patients with unexplained infertility and RSA suffer from polyclonal B-cell activation. Antithyroid antibodies appear to be markers for abnormal T-lymphocyte function. Significant increases in the endometrial T-cell population and the cytokine interferon gamma have been observed in infertile women with ATA. It can be presumed that infertile patients who demonstrate ATA can be classified as having the reproductive autoimmune failure syndrome (RAFS). Patients with RAFS should have immune evaluations that include blocking antibodies, ANA and APA panels, NK cell number and activity, DQ alpha genotyping, and gene mutations leading to inherited thrombophilias. Treatment for Antithyroid AntibodiesIn IVF patients, antithyroid antibodies (ATAs) are treated with intravenous immune globulin (IVIg) before the IVF transfer. There is no specific treatment for ATA in patients with recurrent miscarriage unless it is associated with other abnormalities. Immunophenotypes: Natural Killer Cells and Cytotoxic B-CellsThe immune system is composed of more than 30 types of white blood cells including neutrophils, monocytes and lymphocytes. Lymphocytes, particularly B-cells (antibody producers), T-cells (helper and suppressor) and killer (NK) cells have been the focus of intense research interest to the discipline of reproductive immunology. Immunophenotype refers to the relative amounts of T, B and NK cells in the bloodstream. The immunophenotype assay involves labeling a sample of blood with fluorescent dyes directed to specific markers for each type of lymphocyte: CD4 for T-helper cells, and CD56 for NK cells. The specimen is then placed into a cell flow cytometer in which the cells, via laminar flow fluidics, pass in single file across an argon laser that excites the dyes and causes them to fluoresce. Intensity of the fluorescence is measured by electronic tubes and digitized, allowing a computer to calculate the relative percentages of different lymphocyte subsets. There is a special class of NK cells (CD3-, CD16-, CD56+) in the placenta that promotes cell growth, secretes growth molecules for the placenta and down regulates the mother's immune response locally at the maternal/placenta interface. Opposing is another group of NK cells (CD3-, CD16+, CD56+), when activated by the cytokine IL-2, are cytotoxic to placental trophoblast. The same cells secrete tumor necrosis factor (TNF) which can destroy the placenta. Women with CD16+, CD56+ NK cells in excess of 20% are at risk for miscarriage despite optimal immune treatment (paternal leukocyte immunization, prednisone, aspirin and heparin). In a subset of women who have had multiple failed IVFs, it is believed that TNF is secreted in amounts that inhibit implantation and early formation of the placenta resulting in an IVF cycle which does not produce a clinical pregnancy. Treatment for ImmunophenotypesWomen who have an elevation of NK cells are candidates for immunologlobulin G infusion (IVIg). The dosage of IVIg is 400 mg/kg/day for three consecutive days, monthly, until the NK cells become normal or until the 28th week of pregnancy. In some studies, autoantibodies to phospholipid and nuclear epitopes were demonstrably lower after IVIg. Some researchers have used Enbrel, a TNF alpha inhibitor, instead of, or in addition to IVIg. Presently, there is not enough data to assess the true efficacy of Enbrel therapy. SuccessA recent study reports an 80% success rate in women treated with IVIg who either had a history of miscarriage despite optimal immunotherapy (paternal leukocyte immunization, aspirin, heparin and prednisone), or had a history of IUGR. DQ Alpha GenotypingDQ Alpha genotyping refers to a specific kind of HLA (tissue) typing done at the DNA level. The Class II HLA, found on the surface of white blood cells (WBC), include HLA-DR and HLA-DQ. B-cell is a type of WBC that manufactures antibodies. Each tissue type is made up of an "A" or alpha part and a "B" or beta part. Researchers have discovered that mothers who are HLA-DQ alpha (DQA1) and/or DQ beta (DQB1) compatible with their fetuses tend to have a high rate of miscarriage before eight weeks of pregnancy. Others studies have shown that mother's who are DQ alpha compatible with their fetuses can develop an exacerbation of autoimmune processes, such as rheumatoid arthritis and antiphospholipid antibody syndrome, during the early portions of their pregnancy. Once the association is discovered, more aggressive treatments can be tailored to a patient's situation to prevent the autoimmune process from causing another miscarriage. Inherited ThrombophiliasThe Inherited Thrombophilias comprise a group of genetic disorders of the blood clotting pathways, leading to abnormal blood clot formation (thrombi). A common route involves resistance to a natural anticoagulant called activated protein C (APC). These diseases have been shown in several studies to cause vascular complications that lead to miscarriage, intrauterine fetal death, pre-eclampsia (toxemia of pregnancy), and the HELLP syndrome which is a severe form of pre-eclampsia characterized by hemolysis (blood cells breaking up), elevated levels of liver enzymes, and thrombocytopenia (a low platelet count). Women who carry the genes for Inherited Thrombophilias are more likely (2 to 14 times) to have a clotting problem leading to a miscarriage, compared with the normal population. The three major gene mutations that lead to Inherited Thrombophilias are:
The most common cause of APC resistance arises from the point (one DNA based-pair) mutation at the cleavage site of factor V, called factor V Leiden. It is the most common of the Inherited Thrombophilias, with a prevalence of 10% in the Caucasian population. The mutation has been discovered in 60% of patients who have clot formation during pregnancy, and is also a major cause of blood clots associated with oral contraceptive use. The Prothrombin (factor II) gene mutation has been shown to occur in 7.8% of women who experienced fetal loss due to a clotting disorder. Factor II is one of the major factors in the human clotting pathway. Homocysteine is normally present in low levels in the bloodstream. It is derived from dietary methionine, an amino acid. A gene mutation for the enzyme methylene-tetrahydrofolate reductase (MTHFR), will lead to build up of homocysteine in the bloodstream. This condition, called hyperhomocytseinemia, results in blood clot formation and hardening of the arteries, even in childhood. Nutritional lack of vitamins B6, B12 and folic acid aggravate the problem. Women who have the homozygous form of the MTHFR gene mutation (both of her alleles having the mutation) are more than a two-fold increased risk for a miscarriage. Treatment of Inherited ThrombophiliasA combination of low-dose aspirin plus low molecular weight heparin injections is used to treat the inherited thrombophilias. The therapy starts before pregnancy occurs, and continued four to six weeks after birth. Folic acid supplementation is given to patients with the MTHFR gene mutation. Blocking AntibodiesEarly in pregnancy, the mother's immune system receives signals from the tiny fetus. Many of the signals are hormonal, but others come directly from genetic messages that the father has contributed. Some of the messages involve the tissue type, also known as the human leukocyte antigens (HLA) and the white blood cell (leukocyte) type. HLA are expressed on white blood cells. They are unique to each individual and allow the body to identify anything foreign to it such as infections, cancers, transplanted organs and fetuses. One half of the fetus's HLA type is contributed by mother and the other half by father. When a woman becomes pregnant, her body's immune system usually recognizes the father's HLA as different from her own, and the white blood cells in her uterus produce protective, blocking antibodies. These antibodies coat the baby's cells and protect the fetus from mother's killer cells. If father's HLA is too similar to mother's, her cells may not recognize differences that are vital to the production of blocking antibodies. 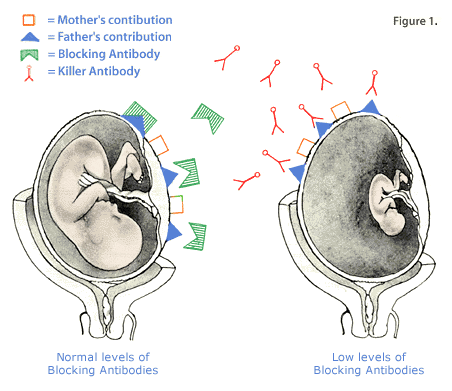
Women who have successful pregnancies and have no history of miscarriages normally, have high levels of blocking antibodies even in the nonpregnant state vs women who miscarry and whose levels tend to be low even when pregnant.
Table 1: Comparison of Blocking Ab level in women with vs. women without RSA
Ab - Antibody RSA - Recurrent spontaneous abortion Through HLA tissue typing we can identify couples who look too much "alike". In addition we can measure the ability of a couple's cells to respond to each other i.e. level of blocking antibodies, using sophisticated equipment which combine computers and lasers (cell flow cytometry). Treatment for Blocking AntibodiesTwo treatments have been offered for low blocking antibody levels: paternal white cell immunizations and IVIg. Immunizing the mother with concentrates of the father's white blood cells amplifies the HLA signal. Although effective, this treatment has not been offered in the U.S. since 2001.
Table 2: Success rate with varying treatment modalities in women with history of RSA
* Medication for APAs and ANAs include low dose aspirin, heparin and prednisone when indicated. ANA - Antinuclear antibodies APA - Antiphospholipid antibodies PLI - Paternal leukocyte immunization IVIg (Intravenous Immune Globulin) is the current therapy. The doses vary between 10 and 60 grams per month. Some doctors have had success with the lower doses of IVIg (10 grams) for patients who have only the blocking antibody problem. The blocking antibody levels should be measured monthly to determine the need for future infusion. Intralipid (a component of TPN) has been successfully used in one small study to prevent miscarriage. There is much interest in the use of Intralipid as it is less costly than IVIg but not enought studies yet exist to whole-heartedly endorse this treatment. The biological mechanism is not known. Laboratory TestingStudies recommend include: Blocking Antibody level (by flow cytometry)T cell IgG B cell IgG Antiphospholipid Antibody Panel Anticardiolipin antibodies -- IgG, IgM, IgA Antiphosphoglycerol antibodies -- IgG, IgM, IgA Antiphosphoserine antibodies -- IgG, IgM, IgA Antiphosphoethanolamine antibodies -- IgG, IgM, IgA Antiphosphatidic acid antibodies -- IgG, IgM, IgA Antiphosphoinositol antibodies -- IgG, IgM, IgA Activated partial thromboplastin time (APTT) Lupus anticoagulant (LA) VDRL Antinuclear Antibody Panel ANA Titer Double stranded DNA SSA SSB RNP SM Antihistone Antibody HLA Tissue Typing ABC DR,DQ DQA1 DNA fingerprinting DQB1 DNA fingerprinting Immunophenotype Natural Killer Cell Activation Assay Natural Killer Cell Activation/IVIg Assay Quantitative Immunoglobulin Factor V Leiden Gene Mutation Factor II (Prothrombin) Gene Mutation Methylene Tetrahydrofolate Reductase (MTHFR) Gene Mutation Written by: William L. Matzner, PhD 
|